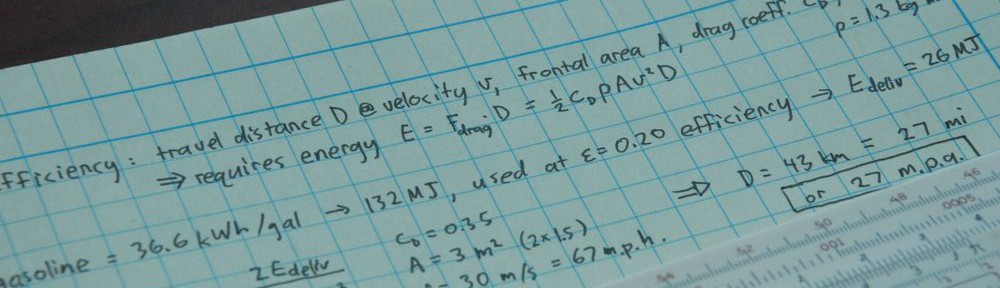
Some themes appear to exert a magnetic pull on my attention: I keep coming back to them and often feel like I’m on a treadmill. It’s hard to figure out why: why am I compelled to keep these topics alive? A recent insight ties some of it together.
Years ago, I wrote a post called A Physics-based Diet Plan. The premise is that humans do not create or destroy atoms within their bodies, and that the energetics are too minuscule to register measurable mass–energy conversion. As such, a person’s mass change—as measured on a bathroom scale, for instance—from one day to the next is completely captured by the difference of mass inputs and mass outputs. It’s just atoms in and out.
Now, the human body has many channels for mass loss. Bathroom functions, breathing (net carbon/water loss really adds up!), and perspiration being the main mechanisms. Mass gain is almost entirely through our pie-holes. And entry via that channel is almost always facilitated by a hand delivering food to the mouth under the control of a brain. If you want to lose weight, the directive is simple: eat less and breathe more. In other words: diet and exercise. I know: radical, right? Every successful program for weight loss involves essentially this same advice, in various guises. That’s because it’s just atoms in every single case, at the foundational level. But oh boy, you wouldn’t believe the resistance I get to this framing. Let’s talk about that…
Continue readingViews: 4979